1. The Recovery Gap: Moving Beyond the Plateau
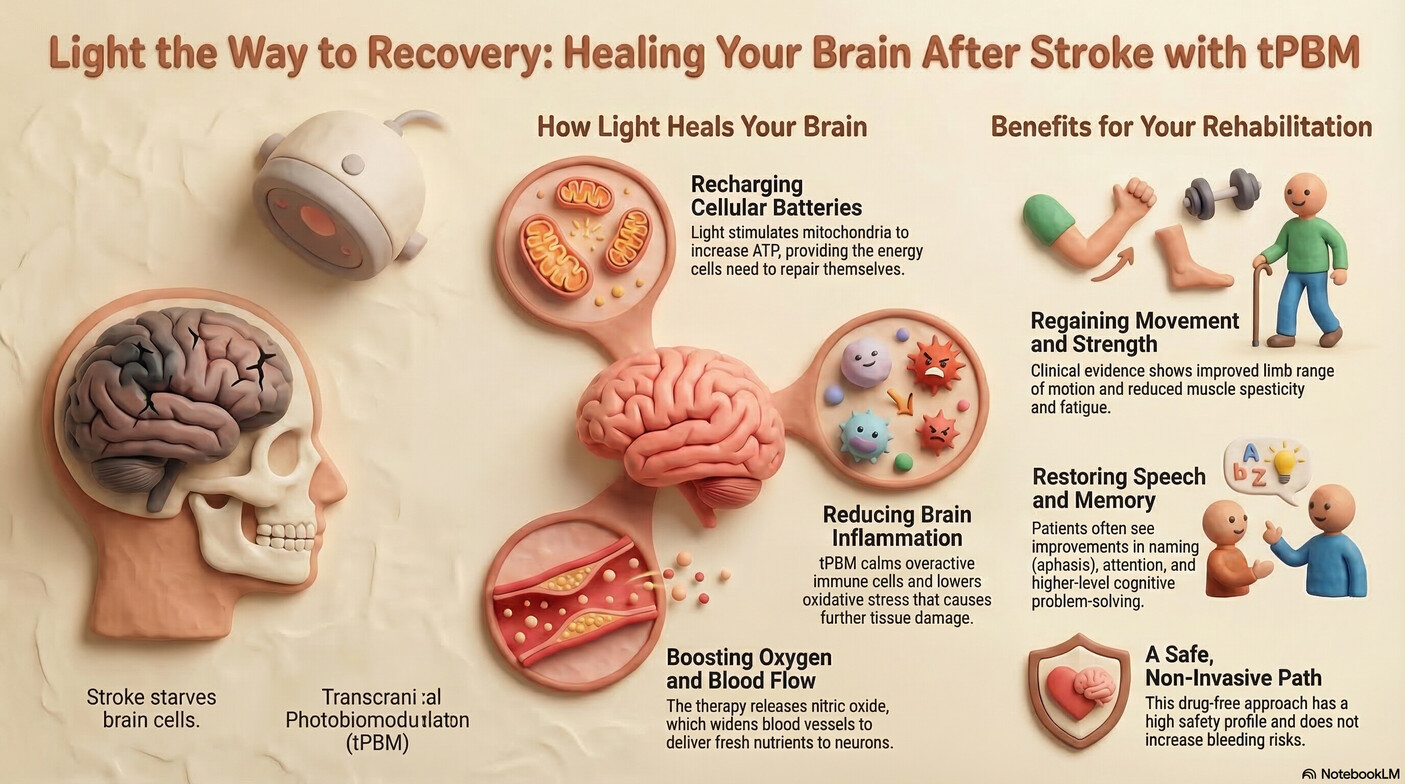
For many survivors of an ischemic stroke, the journey toward recovery eventually hits a frustrating "plateau." After the initial window for traditional interventions—like thrombolysis or surgical thrombectomy—has closed, patients often find that motor and cognitive progress slows. While traditional physical therapy focuses on retraining the brain to move the body, a gap often remains: the biological "fuel" required for the brain to rewire itself is frequently depleted.
As a neuro-rehabilitation specialist, I often see this as a state of chronic neurovascular unit (NVU) dysfunction. Transcranial Photobiomodulation (tPBM) is emerging as a hopeful, non-invasive way to bridge this gap. By using specific wavelengths of red and near-infrared (NIR) light, we can target the brain’s natural repair mechanisms, supporting the "ischemic penumbra"—the salvageable tissue surrounding the initial injury—long after the event.
2. The “Battery” Effect: Recharging Ischemic Neurons
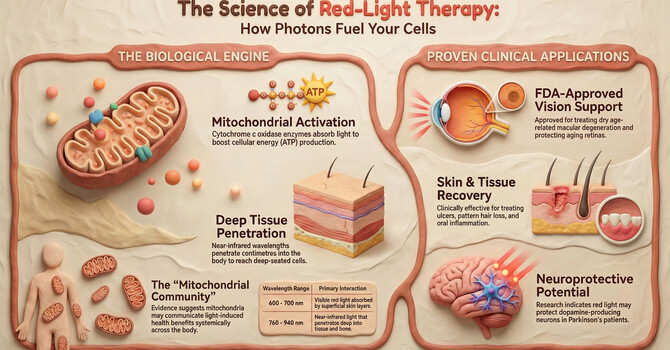
During a stroke, brain cells are starved of oxygen, leading to a catastrophic drop in energy production. In the acute phase, hypoxia causes endogenous Nitric Oxide (NO) to bind to Cytochrome c Oxidase (CCO), the terminal enzyme in the mitochondrial respiratory chain. This bind effectively "locks" the cellular engine, preventing the production of Adenosine Triphosphate (ATP).
tPBM acts as a wireless charger for these cells. When NIR light (600–900nm) reaches the brain tissue, it triggers the photodissociation of NO from CCO. This releases the "brake" on the mitochondria, restoring oxygen consumption and ATP synthesis. Furthermore, advanced research suggests a non-CCO mechanism involving Interfacial Water Layers (IWL). Light at wavelengths like 670nm reduces the viscosity of the water layer near the mitochondrial membrane, allowing the ATP synthase γ-subunit "engine" to rotate more easily.
This is a clinical game-changer. While traditional therapy focuses on using the brain, tPBM focuses on powering it. As the scientific literature notes:
"tPBM targets mitochondrial cytochrome c oxidase to enhance mitochondrial oxidative phosphorylation, increase ATP synthesis... and alleviate energy deficits."
3. The Brain’s “Waste Management” System: Flushing Out the Damage
Recovery requires more than just energy; it requires a clean environment. Stroke leaves behind metabolic "debris," including inflammatory cytokines and red blood cell extravasation. If the brain’s environment remains toxic, neuroplasticity is inhibited.
We now understand that tPBM stimulates the "cleanup crew" of the brain: the Glymphatic and meningeal lymphatic systems. While NIR-I (red/NIR) light supports cellular energy, the burgeoning use of NIR-II wavelengths (specifically 1267nm) targets the Brain Waste Removal System (BWRS) directly. This deeper-reaching light triggers the generation of singlet oxygen, which induces lymphatic vessel relaxation and increases the polarization of Aquaporin-4 (AQP4) at the astrocytic endfeet. This process facilitates the flushing of toxins and shifts the immune microenvironment from a proinflammatory "M1" microglial phenotype to a pro-recovery "M2" phenotype.
4. The "Goldilocks" Principle: Technical Dose Calibration
In neuro-rehabilitation, more light is not always better. We operate under the Arndt-Schulz Law, or the Biphasic Dose Response. This principle dictates a specific "therapeutic window" where light triggers repair.
- Below the Window: The dose is insufficient to trigger a biological response.
- The "Goldilocks" Zone: Low-level light stimulates ATP production and reduces oxidative stress.
- Above the Window: High-energy doses can cause mitochondrial damage or trigger inhibitory RIRR (ROS-induced ROS release).
To ensure precision, specialists are moving toward Einstein (ℯ) standardization. Standard "J/cm²" metrics often fail to account for how many photons actually reach the target tissue after battling a 7–10mm human skull and varying skin melanin levels. Professional dosing accounts for these attenuations to ensure the "ischemic penumbra" receives the exact energy it needs. As researchers state:
"Optimal biological effects are observed within a defined therapeutic window, while excessively high doses can lead to inhibitory/toxic effects."
5. Beyond the Skull: The Systemic and Limb Connection
tPBM is not strictly limited to the head. We are seeing profound results through remote irradiation—applying light to paretic limbs, the abdomen, or even the blood. This triggers a systemic bio-signaling event that modulates the gut-brain axis.
When we treat a weakened limb with 808nm light, we aren't just improving local circulation; we are triggering stem cell migration to the damaged brain and modulating platelet mitochondrial ATP production. Clinically, this "dual-threat" approach—treating the command center (the brain) and the affected periphery—is essential for reducing spasticity, increasing range of motion, and reducing the localized fatigue that often halts progress in the gym.
6. Rewiring the Network: Strengthening Brain Connectivity
Modern science no longer views stroke as just a "hole in the brain." It is a disruption in a network. Stroke breaks the communication lines in critical circuits like the basal ganglia-frontal lobe loop and the salience network.
tPBM improves Functional Connectivity (FC) by repairing these "wires." It helps the brain move away from inefficient "compensatory hyperconnectivity"—where distant regions work overtime to make up for the damage—and toward refined, functional pathways. By increasing metabolic activity in key nodes of the Default Mode Network (DMN), light helps the brain return to a state of efficient, coordinated communication.
7. The Future of Neuro-Physical Therapy: Lessons from NEST-3
The future of stroke rehabilitation lies in targeted, individualized therapy. We have learned much from the challenges of past trials like NEST-3. That trial was terminated after failing to show significant neuroprotection, but the post-mortem analysis is revealing. NEST-3 used a dose of 1.2 J/cm², which we now calculate as only 0.4 ℯ (Einsteins)—a dose likely far too low to penetrate a 7–10mm human skull and reach the penumbra.
Furthermore, NEST-3 used Continuous Wave (CW) light. Today, we favor Pulsed Wave (PW) modes (such as 10Hz or 40Hz). Pulsing allows for higher peak power to reach deeper tissues without causing thermal damage to the scalp, and a 40Hz pulse may even synchronize with gamma rhythms to further modulate neuroimmune responses.
The shift toward precision, individualization, and biological support is here. By giving the brain the energy and the environment it needs to heal, we can push past the plateau.
If we could give your brain cells the energy they need to rebuild, what would your next milestone look like?